Close the Loop Between Quality Events and the Data That Explains Them
Stop chasing context across systems. Uncountable’s QMS links every quality event to the formulation, batch, raw material, and test data behind it—so teams can act faster, learn faster, and prevent repeat failures.

Built for How Quality Teams Actually Work
Uncountable’s QMS isn’t a bolt-on module or a standalone ticket tracker. It’s a quality management system built directly into the platform where your formulation, testing, and production data already lives.
CAPA Management
Initiate, investigate, assign, and verify corrective and preventive actions through configurable stage-gate workflows. Every CAPA links to the formulation revision, batch record, and test data that triggered it.
Nonconformance & Deviation Tracking
Capture and classify deviations with structured investigation templates. Risk-based escalation rules automatically route critical events to the right stakeholders.
Complaint Management
Log external complaints and tie them directly to production batches, formulations, and raw material lots—so the quality team starts the investigation with context, not questions.
Change Control
Manage formulation, process, and material changes with structured impact assessments, multi-level approvals, and automatic post-change quality monitoring tied to the product’s full history.
Document & SOP Control
Author, review, approve, and distribute controlled documents with full version history. Link SOPs directly to the quality events and processes they govern.
Supplier Quality Management
Track supplier performance against incoming material specs, link supplier lot data to downstream deviations, and manage supplier CAPAs within the same system.
Training Assignment & Competency Tracking
Assign training by role, site, process, or product family. Track completion, recency, and competency for the people doing critical work—so your quality system is more than paperwork.
SOP Change → Training Triggers
When an SOP changes, automatically identify impacted roles and require read-and-understand or retraining before execution. Turn document control into prevention, not just versioning.






See how Uncountable's AI platform supports a root cause analysis (RCA)
What Changes When Quality Data Is Connected
Quality failures don’t start with a CAPA. They start as small signals—drift in a process parameter, a supplier lot that behaves differently, a formulation tweak that changes performance. When those signals live in different systems, you only see them when they become a problem.
Without Uncountable QMS:
Engineer manually gathers context from 3–4 systems over 1–2 days
With Uncountable QMS:
Platform instantly surfaces the linked formulation, batch, raw material lot, and recent process changes
Without Uncountable QMS:
Root cause analysis relies on institutional knowledge and ad hoc queries
With Uncountable QMS:
AI correlates the event with similar deviations across products, sites, and time—highlighting probable root causes
Without Uncountable QMS:
Trends are caught only during periodic management review, if at all
With Uncountable QMS:
Platform proactively flags recurring failure modes and cross-site correlations in real time
Without Uncountable QMS:
Formulation or process changes are documented in PLM; quality impact is assessed separately, if at all
With Uncountable QMS:
Change control triggers automatic quality risk assessment linked to the product’s full deviation and test history
Without Uncountable QMS:
Team spends days assembling records from multiple systems into a coherent narrative
With Uncountable QMS:
Full traceability—from quality event to formulation decision to production batch—is available in a single query
Industries we serve – and why it matters
A QMS shouldn’t feel generic… because quality isn’t generic. The right workflows, evidence, and traceability depend on what you make, how you make it, and what you’re accountable for.
Specialty chemicals & materials (polymers, coatings, adhesives, additives)
Tight spec windows, frequent formulation iterations, and complex supplier inputs. Connected data helps teams spot repeat failure modes early and scale consistent quality across sites.
Industrial manufacturing with complex specs & customer requirements
When customer specs drive everything, quality events need to tie back to batches, processes, and changes with a clear, searchable trail, quickly.
Food & ingredient manufacturing
Traceability and speed matter. Quality teams need fast investigations, controlled documentation, and confidence that the right procedures are being followed every time.
Aerospace / defense-adjacent manufacturing
Documentation isn’t overhead. It's the product. Strong change control, training evidence, and end-to-end traceability are non-negotiable.
Life sciences / GxP environments
Validated workflows, strict access control, and audit-ready records are the baseline. A connected system helps teams maintain confidence between audits, not just during them.
Proactive & predictive quality (connected data + AI)
Modern quality teams can’t wait for an out-of-spec result or a customer complaint to learn something is wrong. Proactive and predictive quality comes from connected data: linking every quality event to the formulation revision, batch context, raw material lots, process conditions, and test results that surround it. When that context is unified, analytics and AI can help surface drift, repeat failure modes, and cross-site patterns earlier. Teams can prioritize investigation, intervene sooner, and prevent the next failure rather than documenting the last one.
A
QA Health Monitor dashboards
Bring leading indicators to the surface - open CAPAs, emerging trends, hotspots, and aging - so the team knows where to act first.
B
One data layer across QMS, QC/LIMS, R&D, and PLM
Predictions need context. When quality events and underlying product/process data live together, you can actually learn from history and apply it.
C
AI-assisted analysis
Use connected records to accelerate correlation and pattern discovery, so root cause work becomes less about searching and more about deciding.
D
Preventative controls and validations
Reduce mistakes at the point of capture with governed workflows and checks, so the data stays trustworthy, and teams don’t inherit preventable messes.
E
Traceability, audit logs, and permissions
Give more stakeholders earlier visibility and responsibility, without losing control. Every action stays attributable, reviewable, and ready for audit.
Three Layers of Quality Intelligence
Standalone QMS platforms are databases with workflows. Uncountable’s QMS adds intelligence—because quality improvement requires understanding why failures happen, not just documenting that they did.
Cross-Event Pattern Detection
Find the signal in the noise – Uncountable’s AI scans across all quality events—CAPAs, deviations, complaints, audit findings—to identify patterns that span products, production lines, sites, and time periods. A single out-of-spec result is an incident. The same raw material lot appearing in deviations across three products is a systemic signal.
Predictive Risk Scoring
Act before the next failure – Using historical quality data, formulation changes, and supplier performance trends, the platform scores incoming production batches for quality risk. Flag at-risk runs before they reach final QC. Shift from reactive investigation to proactive prevention.
Why a Connected QMS Changes Everything
Standalone QMS tools require you to build and maintain integrations with your R&D, lab, and production systems. Uncountable’s QMS is natively connected to PLM, LIMS, and R&D data because it’s built on the same platform. Here’s what that means in practice.
Quality ↔ R&D
When a formulation change triggers a downstream deviation, the development team sees it in context. When R&D makes a material substitution, the QMS automatically flags open quality events on the affected product family. No email chains. No blind spots.
Quality ↔ LIMS
QC test results flow directly into quality event investigations. Out-of-spec flags can automatically generate deviations. CoAs, SPC trends, and environmental data are available inside every CAPA—not in a separate system behind a login.
Quality ↔ PLM
Every quality event is linked to the product’s full lifecycle record: formulation revisions, BOMs, scale-up data, and manufacturing specs. Change controls initiated in the QMS update the PLM record. Product and quality histories are one and the same.
Quality ↔ ERP & Instruments
Sync batch records, production schedules, and supplier data with leading ERPs. Pull instrument data directly into quality investigations. Eliminate manual data re-entry and the errors that come with it.
Hero outputs (the results your business runs on)
A connected QMS should do more than track tasks. It should produce clear, audit-ready outputs that leadership can use to make decisions, resulting in faster releases, fewer repeat failures, and less time spent chasing context.
These are the outputs quality teams use every day to show progress, prove control, and prevent what’s coming next.
Hero output (artifact)
- QA Health Monitor (executive dashboard)
- Closed CAPA with effectiveness evidence
- Audit-ready quality package (on demand)
- Early-warning trend / risk report
- Release decision packet / COA-ready evidence
- Controlled change record (impact + approvals + monitoring)
- Data integrity proof (who/what/when + access controls)
KPI / business outcome it moves
- Faster intervention; fewer surprises; smarter prioritization
- Reduced repeat failures; lower cost of poor quality
- Less audit prep time; fewer findings; faster responses
- Prevented failures; fewer line disruptions; fewer complaints
- Faster releases; fewer disputes; stronger customer trust
- Faster safe change; fewer unintended consequences
- Reduced compliance risk; scalable collaboration
Mechanism (how Uncountable enables it)
- Connected events + structured context surfaced in role-based dashboards
- Stage-gated workflows, required evidence, and effectiveness checks
- Traceability + controlled docs + audit logs + approvals in one system
- Connected history enables earlier detection of patterns and drift
- QC results, specs, and approvals tied to the batch and product record
- Change control connected to quality history and post-change monitoring
- Audit trails + permissions + governed workflows
Hero output
KPI
Mechanism
From Audit Scramble to Audit Confidence
Regulatory compliance shouldn’t be a special project. Uncountable’s QMS builds traceability into how your team already works—so the audit trail creates itself.
- Automatic Audit Trails – Every action, approval, escalation, and edit across every quality event is logged with timestamps, user identity, and full context. Nothing to assemble. Nothing to reconstruct.
- Electronic Signatures & Approvals – Configure multi-level approval workflows with electronic signatures that meet FDA 21 CFR Part 11 and EU Annex 11 requirements.
- Controlled Document Management – SOPs, work instructions, and quality policies are versioned, reviewed, approved, and distributed within the same platform. No shared drives. No version confusion.
- Global Harmonization – Standardize quality event workflows, escalation criteria, and reporting across all sites. Allow for site-level customization within a governed global framework.
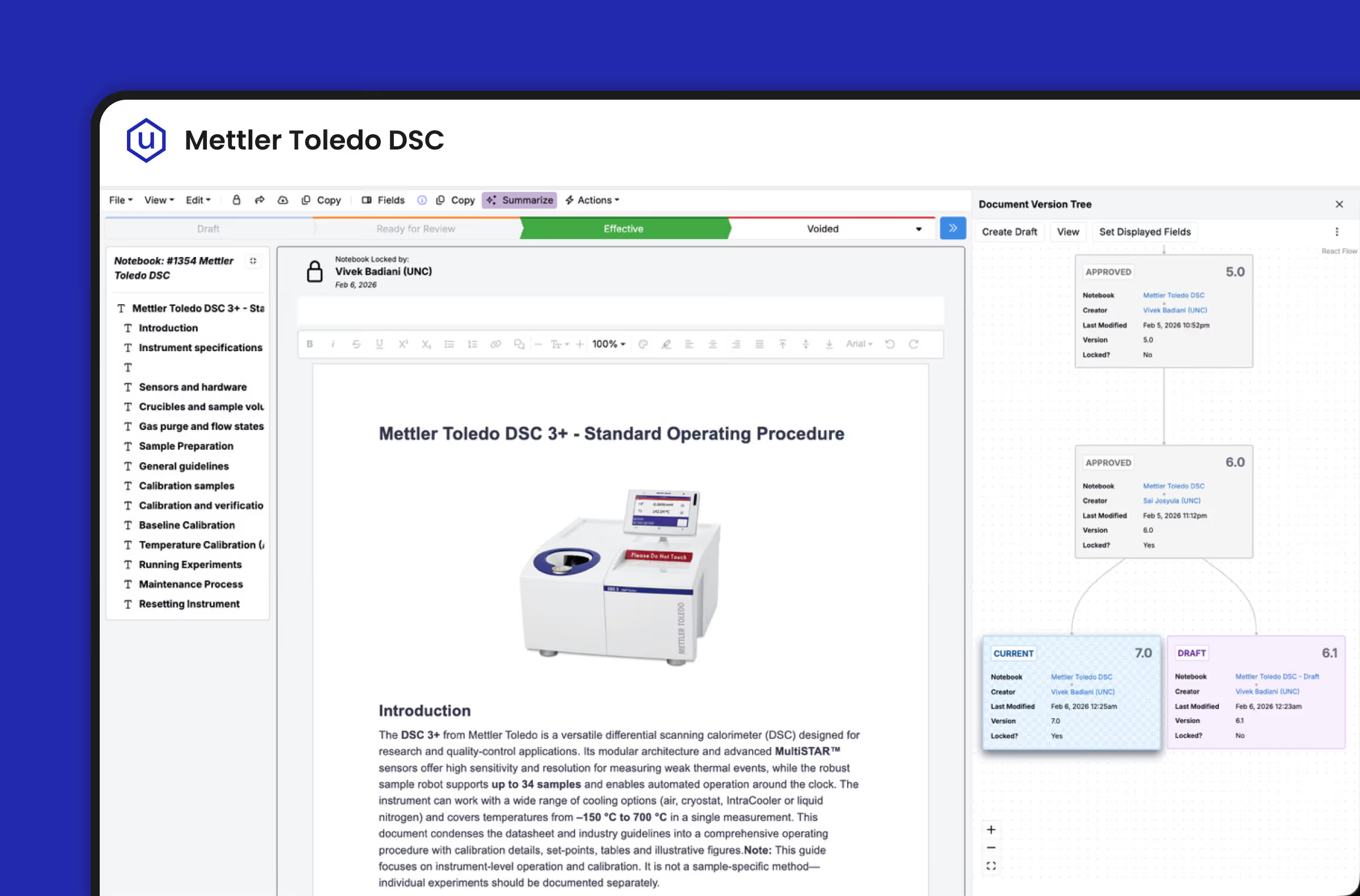
Built for Every Stakeholder in the Quality Chain
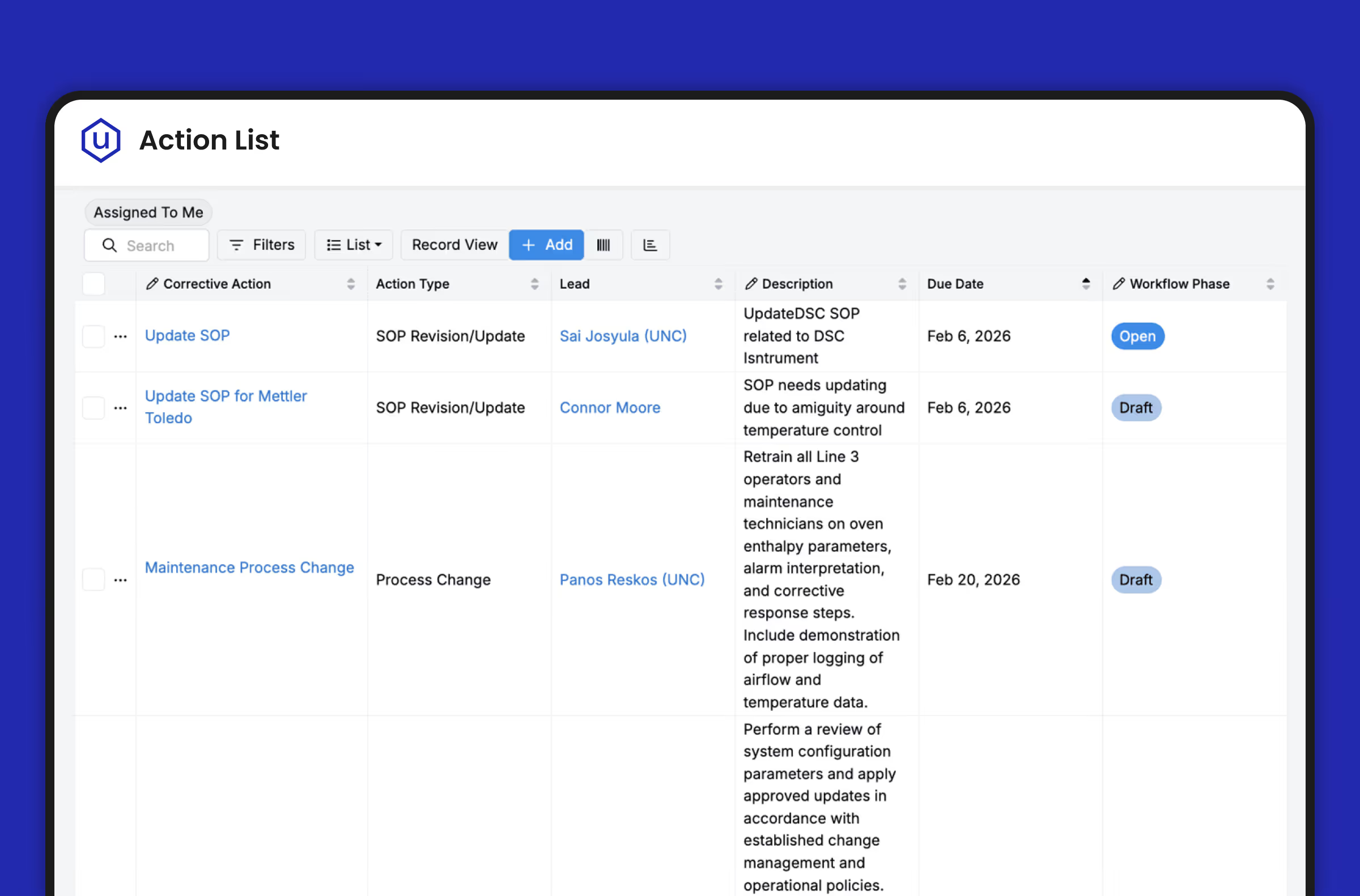
- VP of Quality – Executive dashboards with real-time visibility into open CAPAs, deviation trends, CAPA effectiveness rates, and audit readiness scores across all sites. Management review data is always current—never a quarterly compilation exercise.
- Quality Engineer – Investigation workbenches with every piece of context—formulation history, batch records, raw material data, SPC charts—already linked to the quality event. Spend time analyzing, not assembling.
- Regulatory & Compliance – Audit-ready traceability from any quality event to the underlying product, process, and supplier data. Generate compliance packages in minutes. Maintain confidence between audits, not just during them.
- R&D / Formulation – Visibility into how formulation decisions affect downstream quality. See which material substitutions, process changes, or scale-up adjustments triggered deviations—and use that insight to make better decisions going forward.
- Plant / Operations – Real-time quality dashboards, automated deviation capture from production data, and clear escalation paths that keep the line running while quality events are investigated.
Deploying a QMS in a Regulated Environment
Every Uncountable QMS deployment follows a structured, validation-ready methodology designed for regulated manufacturing. We’ve rolled out quality systems across single-site operations and multi-plant global companies—and every deployment is scoped around your specific compliance requirements, not a generic template.
Map current quality workflows, event types, escalation paths, approval chains, and regulatory requirements. Define what migrates, what changes, and what stays.
Set up quality event types, CAPA stage-gate workflows, approval routing, role-based access, and integrations with your LIMS, PLM, and ERP. Migrate and validate historical quality data.
Run structured test scenarios using real quality events. Confirm workflows, permissions, calculations, and integrations. Your team signs off before go-live.
Operate both systems simultaneously. Your validation team confirms audit trails, electronic signatures, and data integrity meet regulatory requirements before cutover.
Full cutover with embedded support. Direct access to Uncountable’s implementation team for real-time troubleshooting during the first weeks of live operation.
Extend to additional sites, enable AI analytics, refine workflows, and build custom dashboards as your team matures on the platform.
Ready to Connect Quality to the Data That Matters?


